
What combination of chemical elements is your MBTI type? xNFJ (Fe-Ni and Ni- Fe) - Nickel and Iron... | VK

Atomic Cation‐Vacancy Engineering of NiFe‐Layered Double Hydroxides for Improved Activity and Stability towards the Oxygen Evolution Reaction - Peng - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Reactive Fe-Sites in Ni/Fe (Oxy)hydroxide Are Responsible for Exceptional Oxygen Electrocatalysis Activity | Journal of the American Chemical Society

The synergistic effect between Ni sites and Ni-Fe alloy sites on hydrodeoxygenation of lignin-derived phenols - ScienceDirect
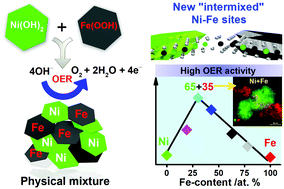
Formation of unexpectedly active Ni–Fe oxygen evolution electrocatalysts by physically mixing Ni and Fe oxyhydroxides - Chemical Communications (RSC Publishing)

Recent Progress on NiFe‐Based Electrocatalysts for the Oxygen Evolution Reaction - Zhao - 2020 - Small - Wiley Online Library
The order of my cognitive functions goes: Ni, Ne, Ti, Fi, Fe, Te, Se, Si. What do you think my meyer-Briggs type is? - Quora
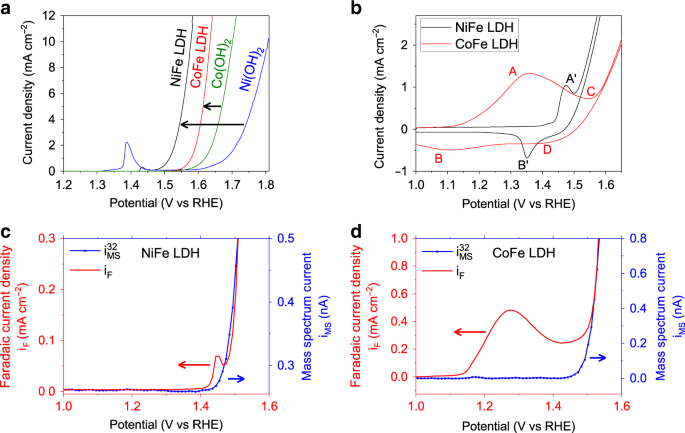
In-situ structure and catalytic mechanism of NiFe and CoFe layered double hydroxides during oxygen evolution | Nature Communications

Further Insight into the Conversion of a Ni–Fe Metal–Organic Framework during Water-Oxidation Reaction | Inorganic Chemistry

Ni-fe Nickel Iron Nickel Iron Batteries 1.2v Iron Nickel Battery 1200ah - Buy Iron Nickel Battery,1.2v 1200ah Battery,1.2v Nife Battery Product on Alibaba.com

Synergy between Fe and Ni in the optimal performance of (Ni,Fe)OOH catalysts for the oxygen evolution reaction | PNAS
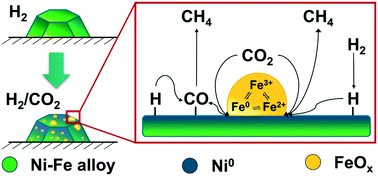




![The Fe-Ni phase diagram from a literature [26]. | Download Scientific Diagram The Fe-Ni phase diagram from a literature [26]. | Download Scientific Diagram](https://www.researchgate.net/publication/272879847/figure/fig2/AS:615086280413192@1523659405670/The-Fe-Ni-phase-diagram-from-a-literature-26.png)
