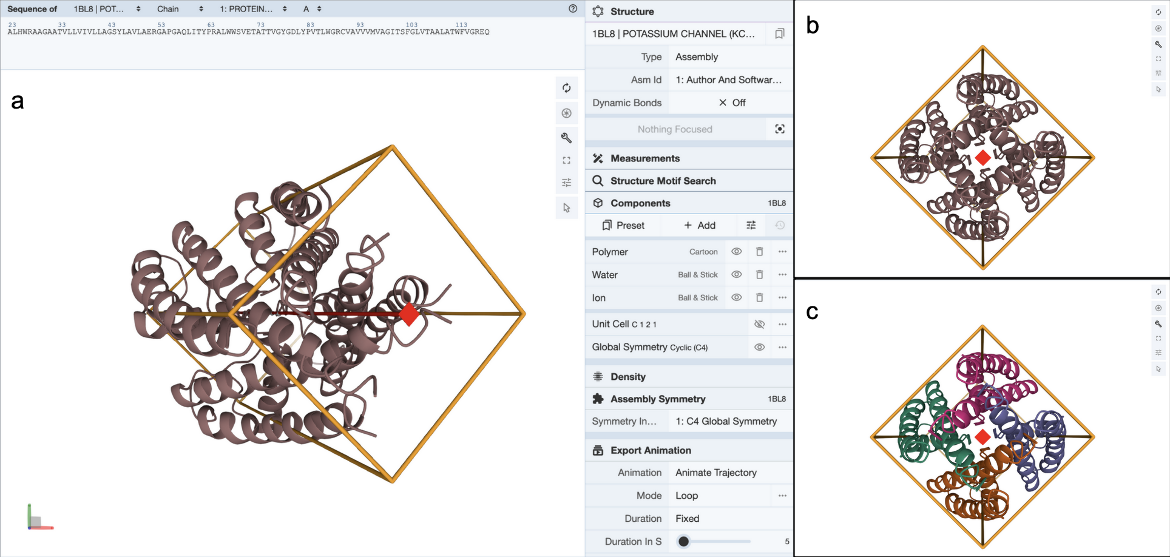
1) At 2257 K and 1.00 atm total pressure, water is 1.77 per cent dissociated at equilibrium by way of the reaction 2 H2O(g) <
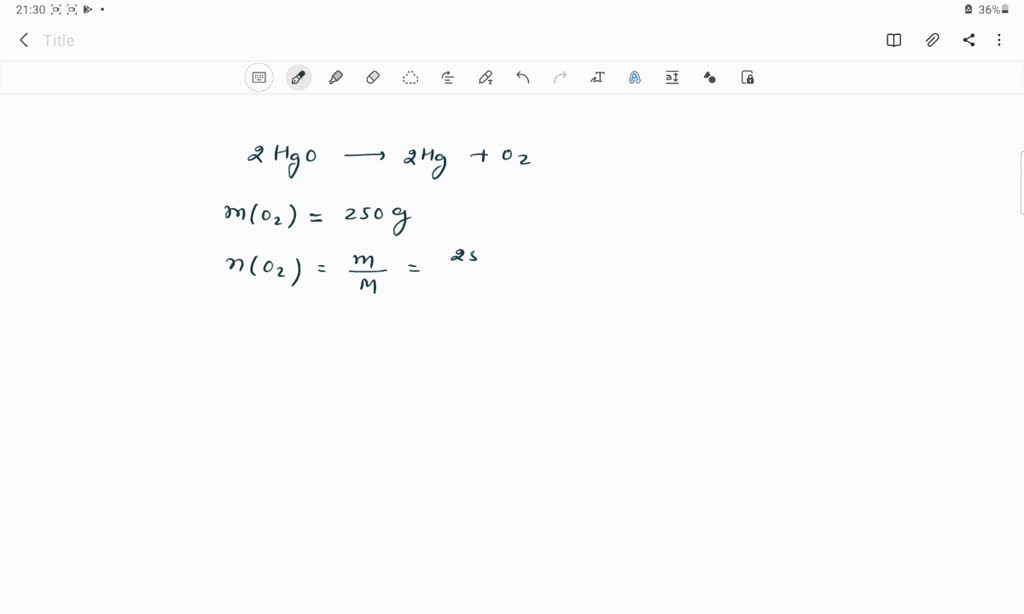
SOLVED: The molar mass of HgO is 216.59 g/mol. The molar mass of O2 is 32.00 g/mol. How many moles of HgO are needed to produce 250.0 g of O2?