Kamil @Nerdabetic (he/him/his) on Twitter: "Libre 3 was submitted to FDA on 20/12/2021; approval received 26/05/2022. Public announcement on 31/05/2022. This clearance means Libre 3 in its current version can't be integrated

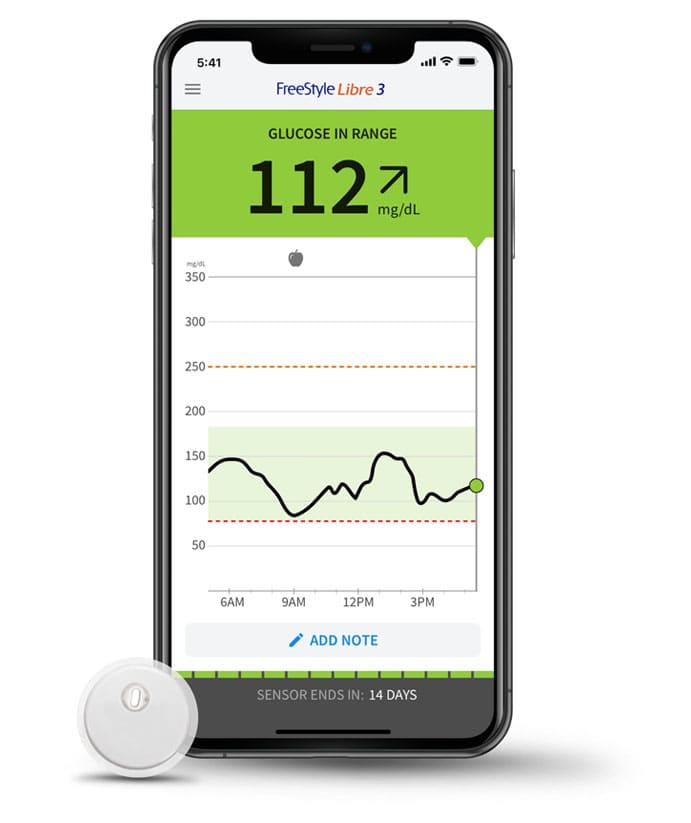
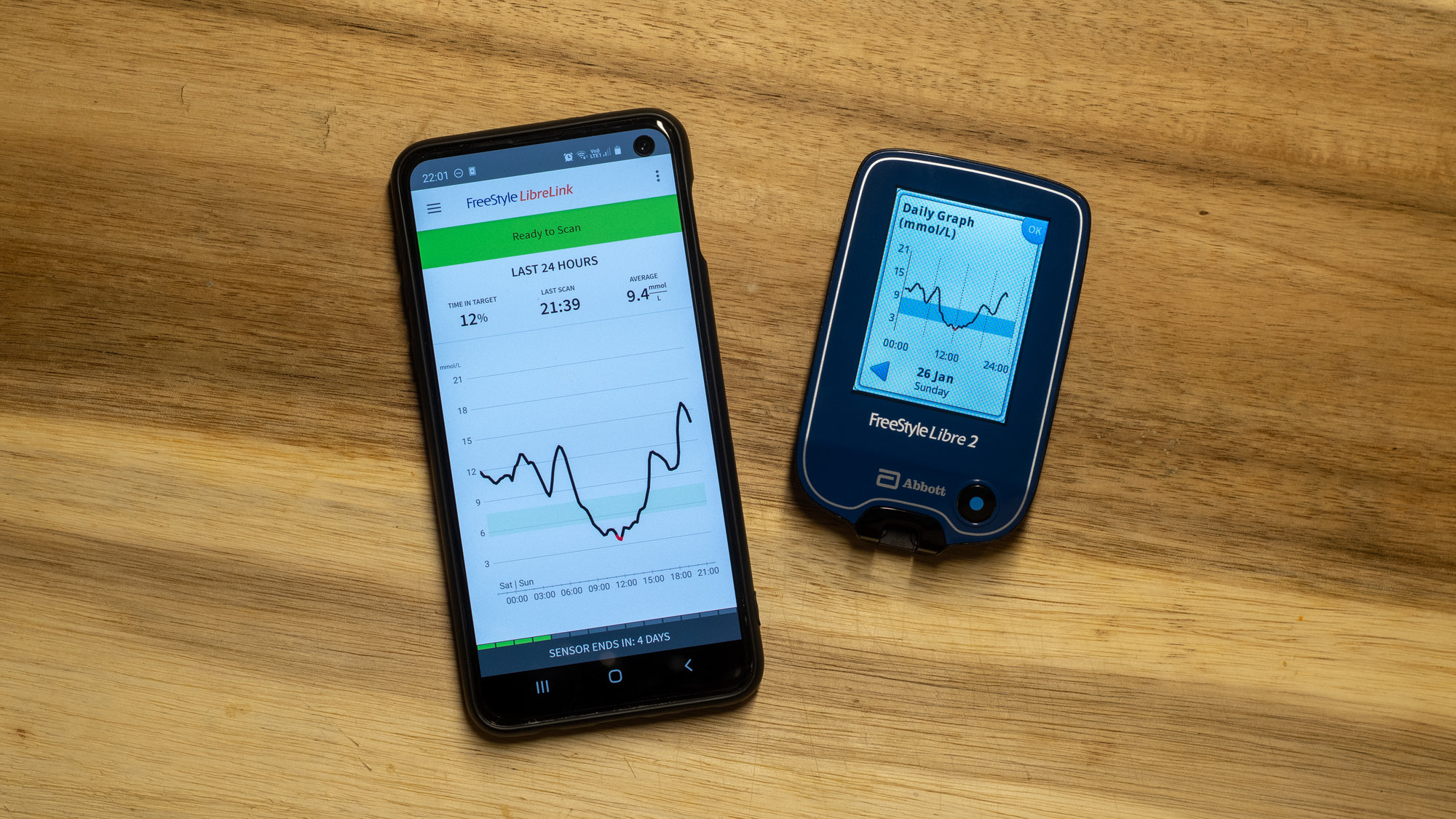
I can't has cheezburger | The Freestyle Libre 3 Puts the “Continuous” in “Continuous Glucose Monitor”
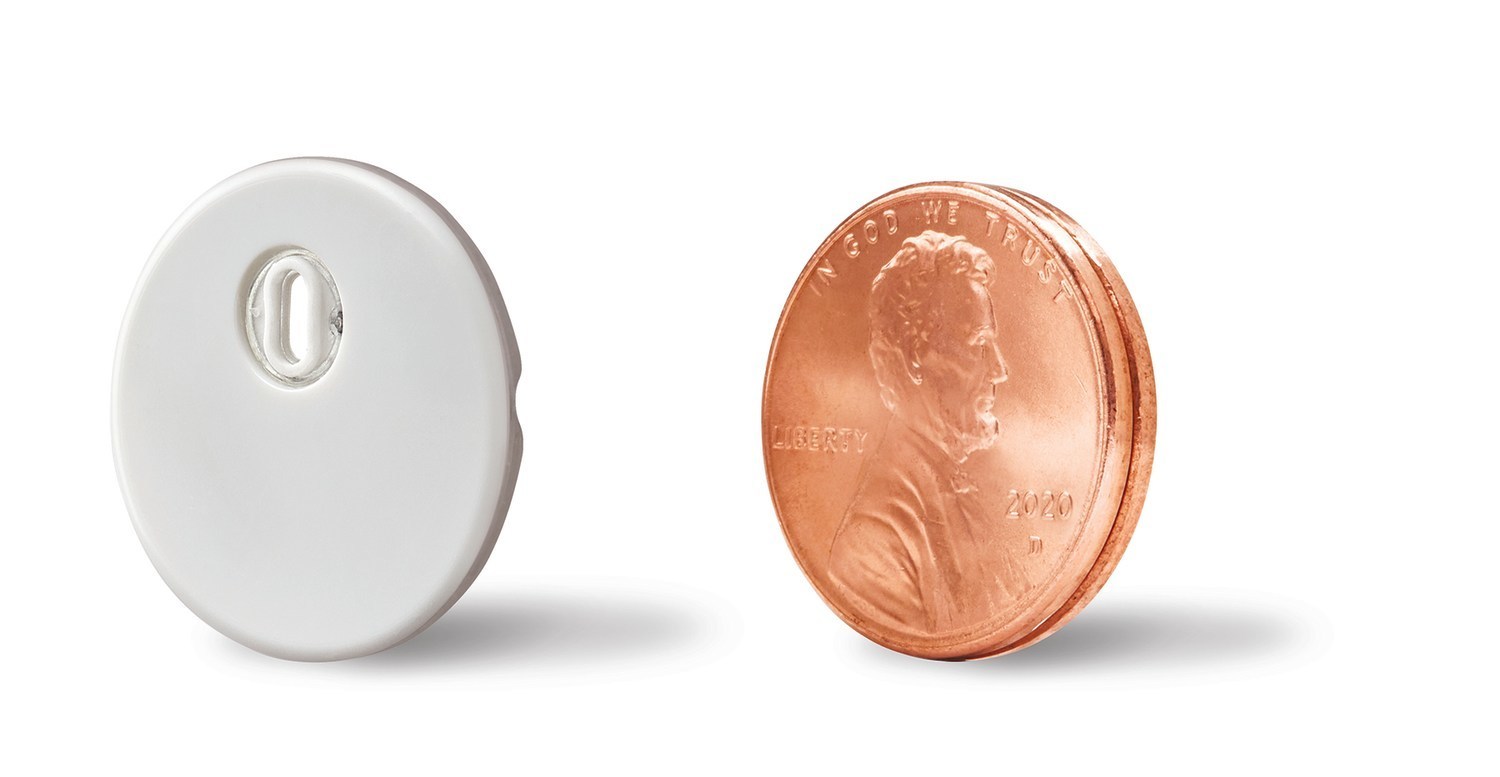
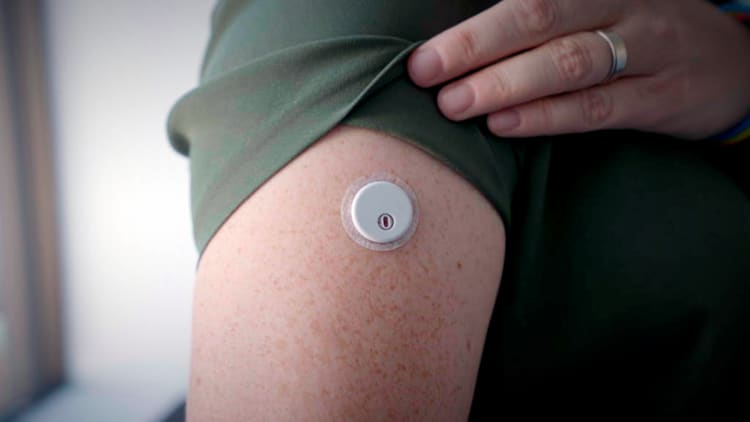


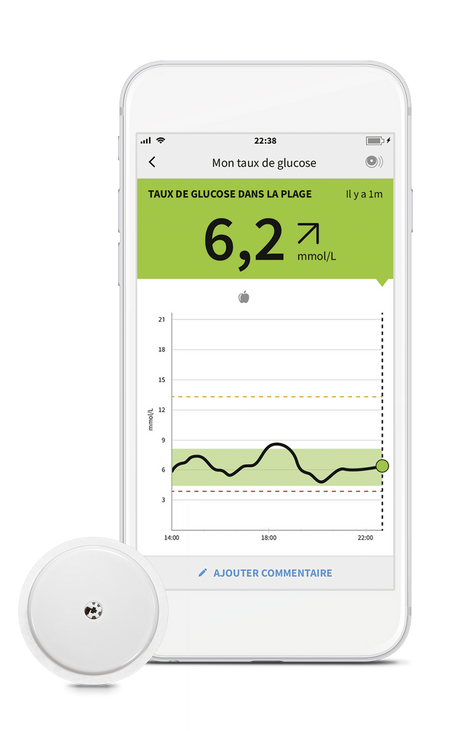

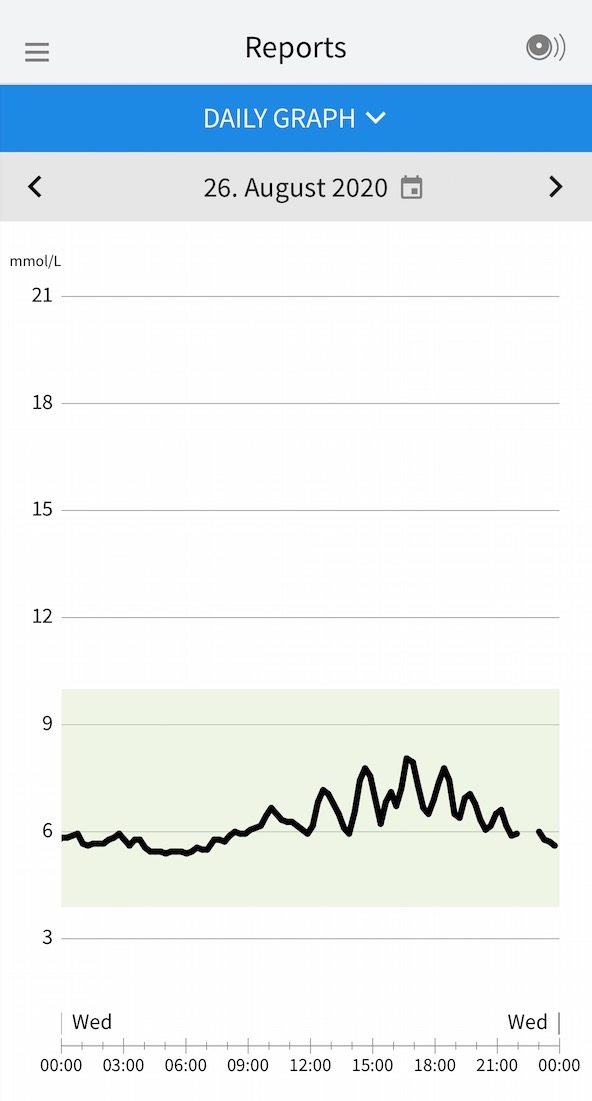


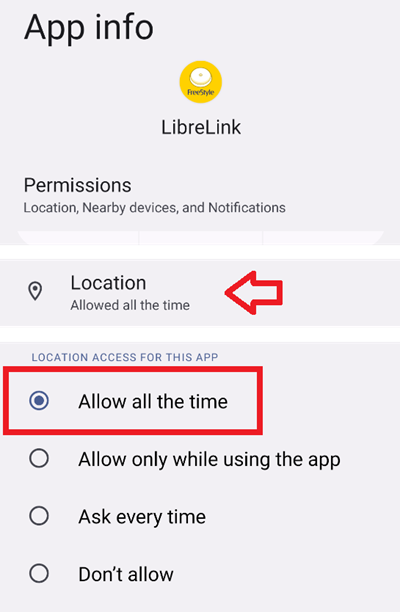

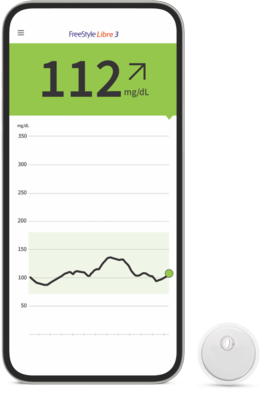

![Freestyle Libre 3 Sensor Buy online - [New Release 2023] Freestyle Libre 3 Sensor Buy online - [New Release 2023]](https://cgmmonitors.com/wp-content/uploads/2022/08/freestyle-libre-3.png)