
SOLVED: Calculate the equilibrium constant Keq; at 25 degrees € for the reaction: 2Br-(aq) 12(s) Br2() 21-(aq). (Eored,cathode=0.54V and Eored,anode=1.066V) Puan) 1.I1xlo 1.62r0" 2.25x10-16 1.52rlO" 1.25rl0

R.I.C.E. Table in Chemistry Formula & Method | How to Calculate Keq - Video & Lesson Transcript | Study.com
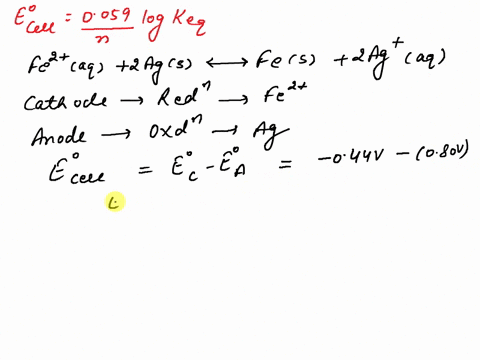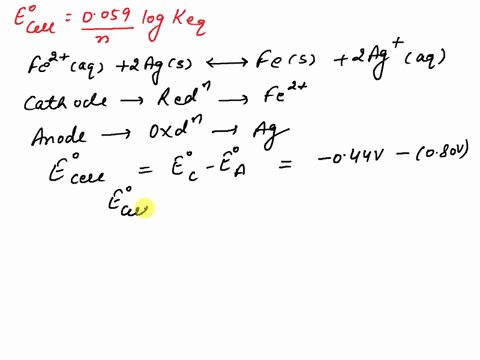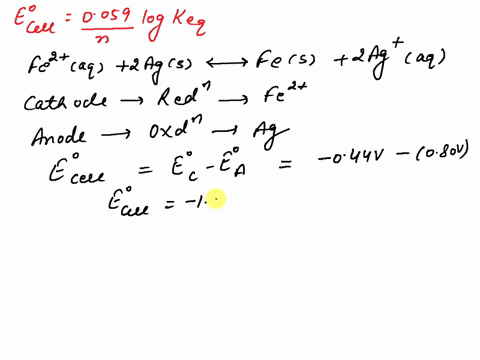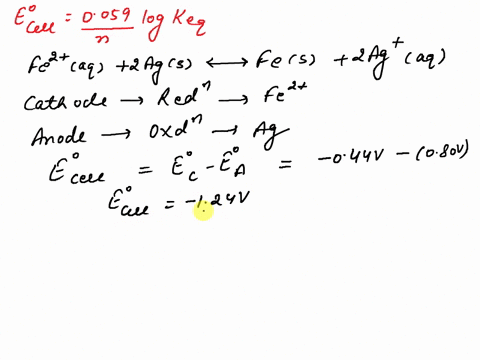
SOLVED: What is the value of the equilibrium constant, Keq, for the reaction between copper metal and iron(III) ions in aqueous solution at 25 ⁰C. EoCu3+/Cu = -0.337V; EoFe3+/Fe2+ = 0.77 V
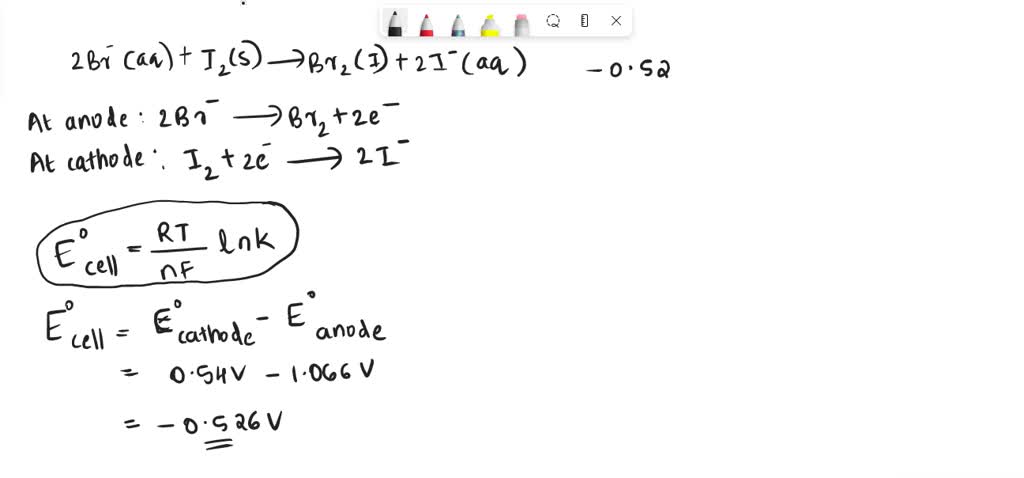
SOLVED: Calculate the equilibrium constant Keq; at 25 degrees € for the reaction: 2Br-(aq) 12(s) Br2() 21-(aq). (Eored,cathode=0.54V and Eored,anode=1.066V) Puan) 1.I1xlo 1.62r0" 2.25x10-16 1.52rlO" 1.25rl0

R.I.C.E. Table in Chemistry Formula & Method | How to Calculate Keq - Video & Lesson Transcript | Study.com
![SOLVED: 0.4 points What is the correct equilibrium constant expression for this reaction Agt (aq) CI- (aq) = AgCl (s) Keq [AgtH[Ct Keq [Ag+] . [CI ] Keq [AgC] [Ag' H[ct-] [Agt ] [ SOLVED: 0.4 points What is the correct equilibrium constant expression for this reaction Agt (aq) CI- (aq) = AgCl (s) Keq [AgtH[Ct Keq [Ag+] . [CI ] Keq [AgC] [Ag' H[ct-] [Agt ] [](https://cdn.numerade.com/ask_images/1f592b4d27ff409f8e5c5a32863d0106.jpg)











