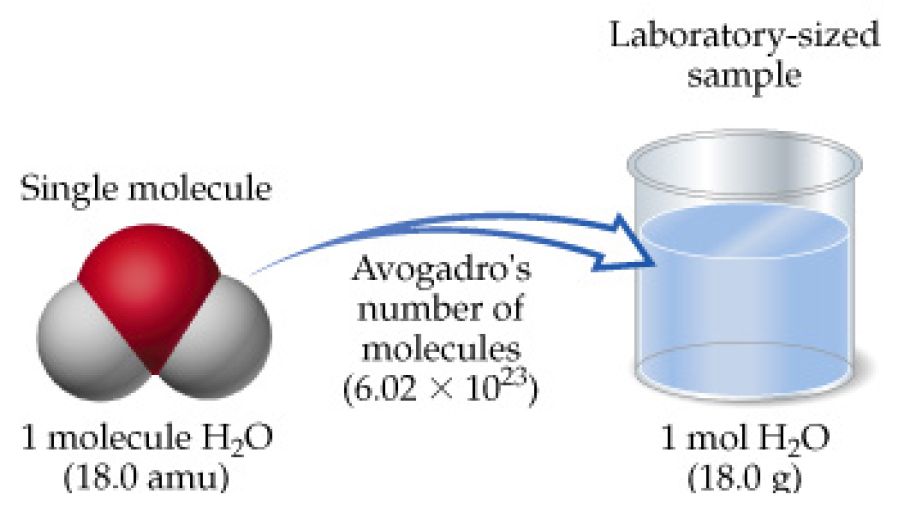
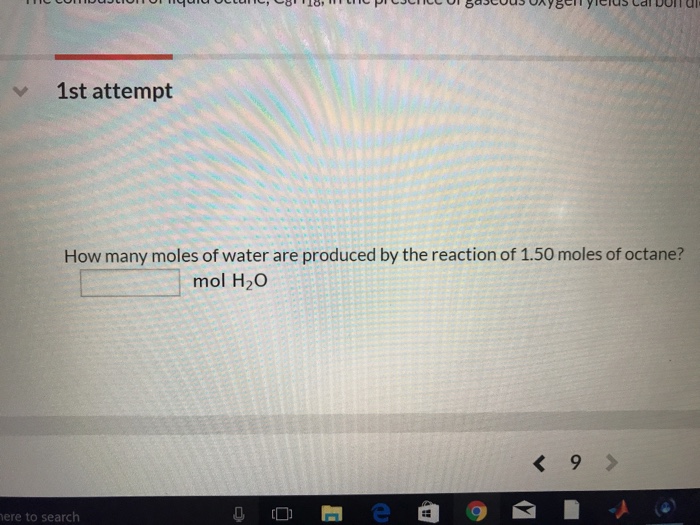
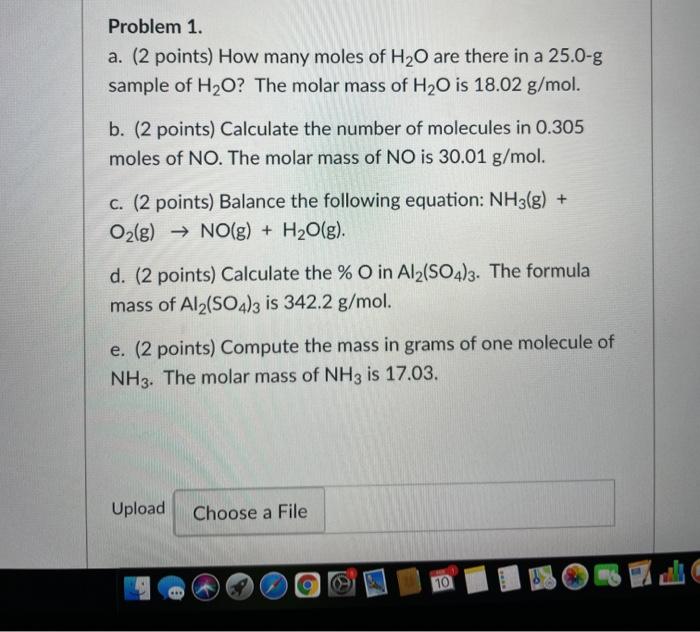
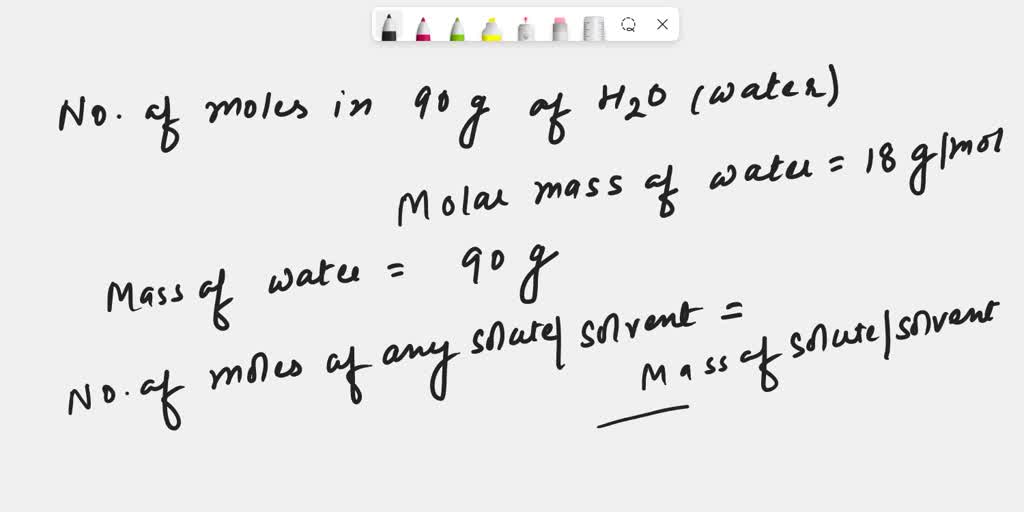SOLVED: 1) How many molecules of H2O are in 1.25 moles of H20? 2) How many grams of He are in 9.84 x 10^22 atoms of He? 3) How many moles of

The molar mass of H2O is 18.01 g/mol . The molar mass of 02 is 32.00 g/mol What is the mass of H20 in - Brainly.com