
SOLVED: QUESTiON 5 In the following reaction CaSO4*2HzO(s) CaSO4(s) + H2O(g) the anhydrate is CaSOa(s) HzO(g) CaSO4 2H2O(s) none of these

Calcium sulfate structure. (a) In gypsum (CaSO 4 ·2H 2 O), the water... | Download Scientific Diagram

Reaction Characteristics of CaSO4/CaSO4·1/2H2O Reversible Reaction for Chemical Heat Pump | Semantic Scholar

Arsenic speciation in synthetic gypsum (CaSO4·2H2O): A synchrotron XAS, single-crystal EPR, and pulsed ENDOR study - ScienceDirect

Thermodynamic Modeling of Calcium Sulfate Hydrates in the CaSO4–H2O System from 273.15 to 473.15 K with Extension to 548.15 K | Journal of Chemical & Engineering Data



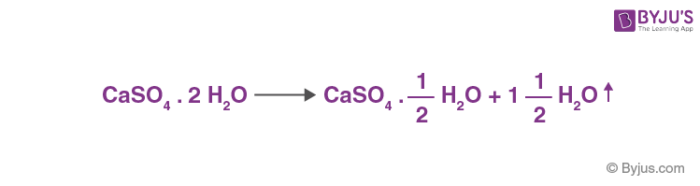

![Y []Δ,205^∘C CaSO4.2H2O []Δ,120^∘C X . X and Y are respectively . Y []Δ,205^∘C CaSO4.2H2O []Δ,120^∘C X . X and Y are respectively .](https://dwes9vv9u0550.cloudfront.net/images/4281429/89baf354-9f4e-4244-93da-acebcca3fce9.jpg)










