Dehydration Pathways of Gypsum and the Rehydration Mechanism of Soluble Anhydrite γ-CaSO4 | ACS Omega

Direct synthesis of single-phase α-CaSO4·0.5H2O whiskers from waste nitrate solution - ScienceDirect
![PDF] Solubility Measurements in the System CaSO4–NaCl–H2O at 35°, 50°, and 70°Cand one atmosphere Pressure | Semantic Scholar PDF] Solubility Measurements in the System CaSO4–NaCl–H2O at 35°, 50°, and 70°Cand one atmosphere Pressure | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f28e8097270f31a666cc3ba8a4dc4d5c500648f8/22-Table6-1.png)
PDF] Solubility Measurements in the System CaSO4–NaCl–H2O at 35°, 50°, and 70°Cand one atmosphere Pressure | Semantic Scholar

Gypsum (calcium Sulfate Dihydrate, CaSO4.H2O) Mineral, Chemical Structure Stock Illustration - Illustration of mortar, drywall: 188464863
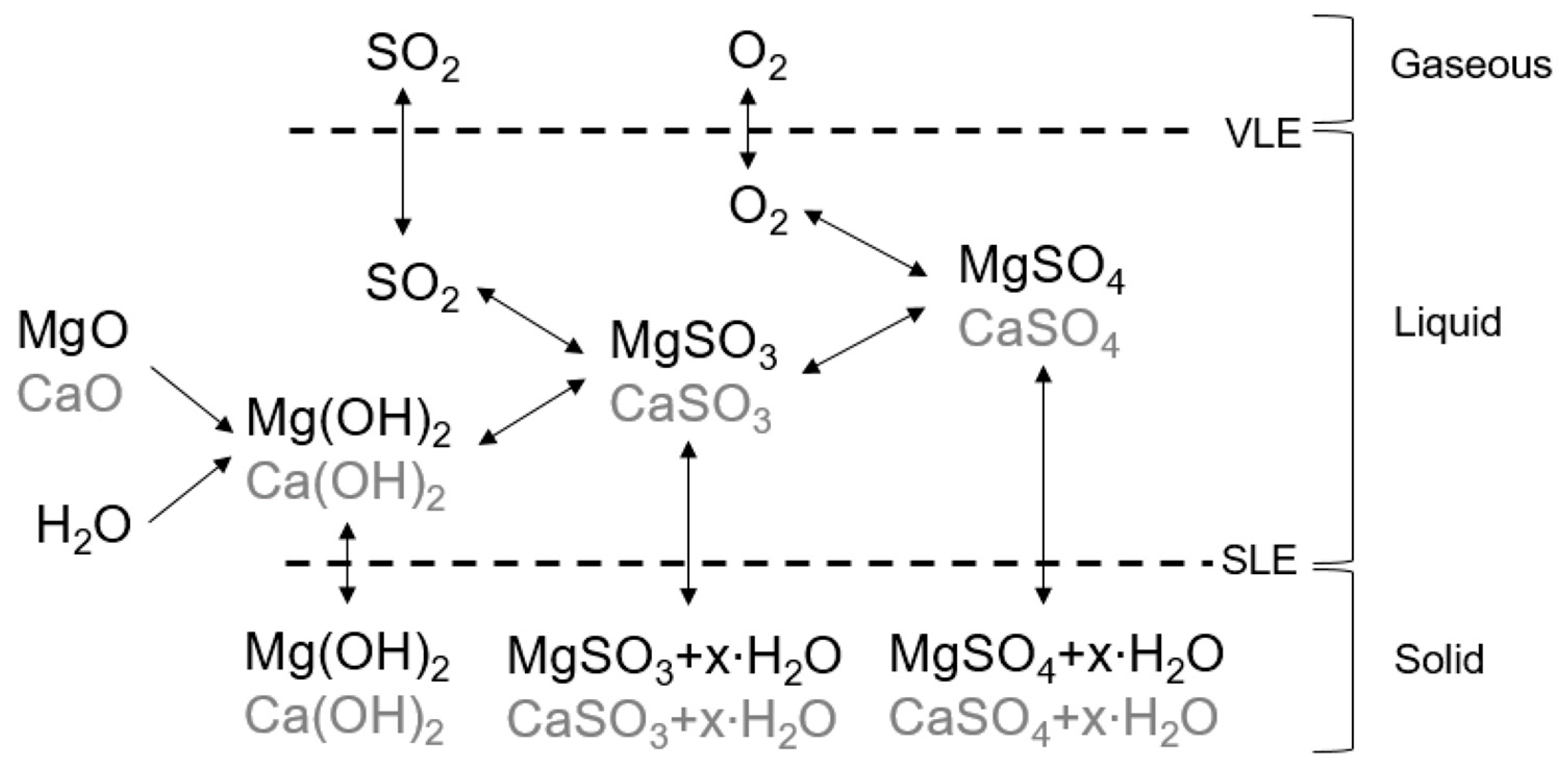
Processes | Free Full-Text | Solubility Data of Potential Salts in the MgO-CaO-SO2-H2O-O2 System for Process Modeling

Complete the following equations:(a) 2NaOH + Cl2→ NaCl + + H2O (b) Na2SO3 + 2HCl→ 2NaCl + H2O + (c) Cl2 + 3F2 573K .

SOLVED: Ca504 HzO(s Caso4 s) + 2 H2o(a) The hyclrale Ca524 2H2o(s canbehealed farn Int #nhvor u* salt CaSOAls) shjynby thic rtaclic" camcGcnro douvt Using lhe dala lable bclow calculale Inc vallic