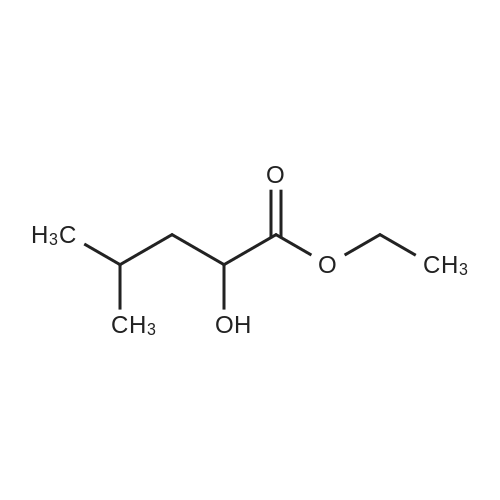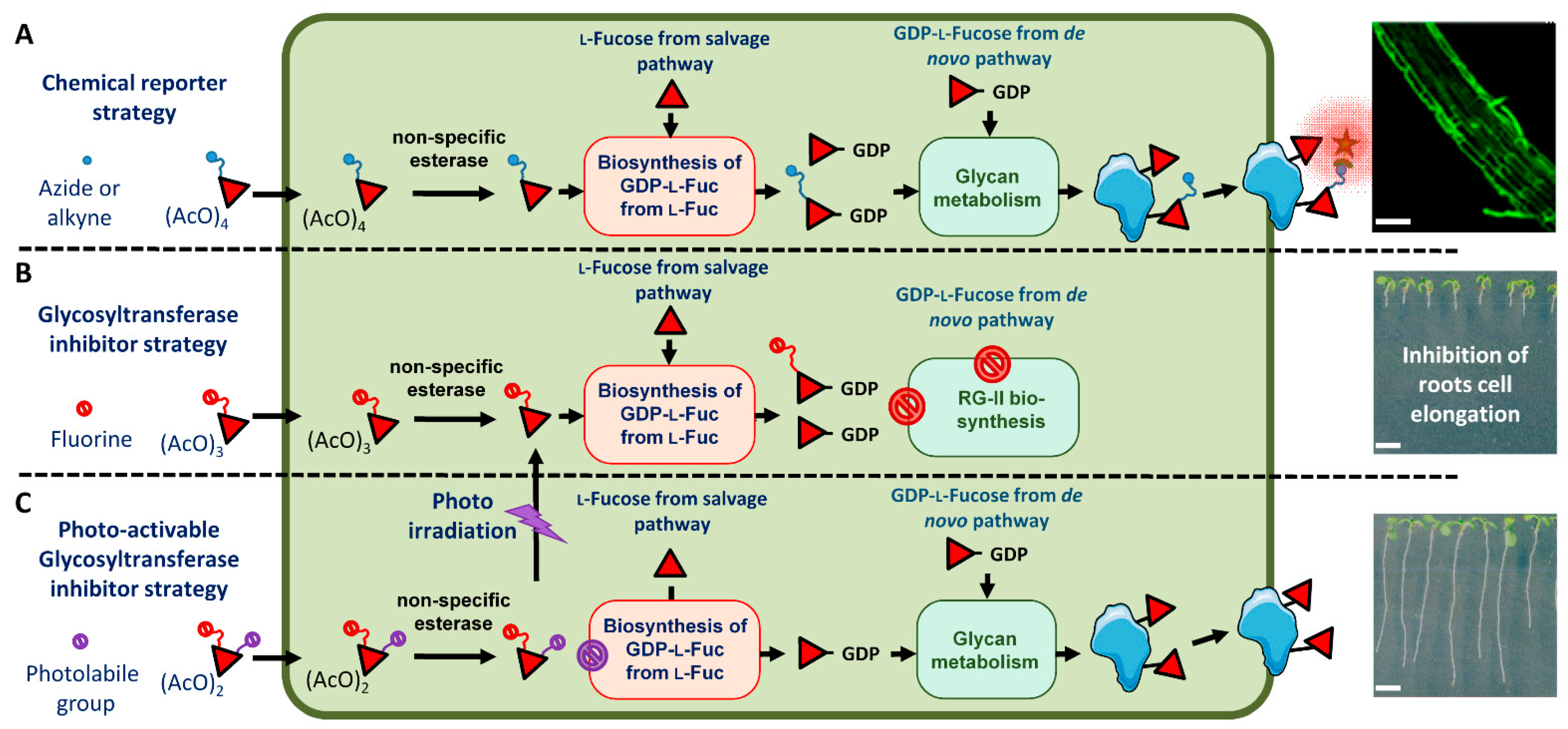
IJMS | Free Full-Text | The Light-Controlled Release of 2-fluoro-l-fucose, an Inhibitor of the Root Cell Elongation, from a nitrobenzyl-caged Derivative

Total Synthesis of (−)-Platensimycin, a Novel Antibacterial Agent | The Journal of Organic Chemistry

Flow Chemistry for Cycloaddition Reactions - García‐Lacuna - 2020 - ChemSusChem - Wiley Online Library
![Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development](https://pubs.acs.org/cms/10.1021/acs.oprd.2c00248/asset/images/large/op2c00248_0016.jpeg)
Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development

Women's NWT Shoes - Clothes Mentor" - cartname-clothes-mentor-minnetonka-minnesota - cartname-clothes-mentor-minnetonka-minnesota

Enantiospecific total synthesis of indole alkaloids (+)-eburnamonine, (−)-aspidospermidine and (−)-quebrachamine - ScienceDirect